1. Pasteurisation is related to
A. sterilisation of milk
B. dehydration of milk
C. fermentation of milk
D. distillation of milk
Pasteurisation is a process in which milk is treated with mild heat to eliminate pathogens and extend the shelf life of milk.
2. Which of the following is not correctly matched?
A. Desalination - Sea water
B. Reverse osmosis - Potable water
C. Denaturation - Proteins
D. Pasteurisation - Tea
Option (d) is not correctly matched because pasteurisation is the partial sterilisation of a product, such as milk, wine or fruit juice to improve their keeping quality.
3. Phosphatase test is used for the analysis of
A. milk
B. tea
C. water
D. All of these
The Alkaline Phosphatase (ALP) test is used to indicate whether milk has been adequately pasteurised or whether it has been contaminated with raw milk after pasteurisation.
4. For a given liquid, the rate of evaporation depends on
I. the temperature of the liquid
II. the temperature of air
III. the surface area of the liquid
Select the correct factor/factors using the codes given below.
A. I only
B. I and II only
C. II and III only
D. I, II and III
For a given liquid, the rate of evaporation depends on the temperature of the liquid, air and the surface area of the liquid.
Higher temperatures increase the kinetic energy of the liquid molecules, leading to a higher rate of evaporation. The temperature of the surrounding air also significantly affects the rate of evaporation. Specifically, higher air temperatures can increase the rate of evaporation. A larger surface area allows more molecules to escape from the liquid, increasing the rate of evaporation.
5. Consider the following statements.
Assertion (A) The temperature below 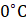 is obtained due to a mixture of salt and ice.
is obtained due to a mixture of salt and ice.
Reason (R) Salt increases the freezing point of ice. IAS (Pre) 1993, UP Lower Sub (Pre) 1998
A. Both A and R are true and R is the correct explanation of A .
B. Both A and R are true but R is not the correct explanation of A .
C. A is correct but R is incorrect.
D. A is incorrect but R is correct.

6. Solutions are of many types. Consider the following types of solution with an example each.
Assam PCS (Pre) 2024
I. Gas in liquid - Vinegar
II. Solid in liquid - Brine
III. Solid in solid - Brass
IV. Liquid in Liquid - Coca-cola
Select the correctly matched pairs using the codes given below:
A. I and II only
B. II and III only
C. I, III and IV only
D. II, III and IV only
Pairs II and III are correctly matched.
Vinegar is a combination of water and acetic acid, often with other flavourings or additives depending on the type of vinegar. So, in a way, vinegar is a liquid within a liquid, with water being the primary solvent. Hence, pair (I) is incorrectly matched.
Brine (or briny water) is water with a high-concentration solution of salt (typically sodium chloride or calcium chloride). Hence, pair (II) is correctly matched.
Brass can be described as a solid in solid, referring to its homogeneous structure where one solid material (copper) is mixed with another solid material (zinc) to form a single solid substance (brass). Hence, pair (III) is correctly matched.
Coca-cola is carbonated water. It is gas in liquid.
Hence, pair (IV) is incorrectly matched.
7. Consider the below two statements.
Statement I Lyophilic sols are more stable than lyophobic sols.
Statement II Lyophilic colloids are extensively solvated.
From the above statements, choose the correct answer from the options given below.
A. Both Statement I and Statement II are true.
B. Statement I is true but Statement II is false.
C. Both Statement I and Statement II are false.
D. Statement I is false but Statement II is true.
Both Statement I and Statement II are true.
The stability of lyophilic sols is a result of two factors. First, the presence of a charge and second, the solvation of colloidal particles. Due to extensive solvation of lyophilic colloidal sols, which form a protective layer outside it which prevents it from forming associated colloids. On the other hand, stability of lyophobic sols is only because of the presence of a charge.
8.With the progress of any chemical reaction, the rate of the chemical reaction ...... with time.
A. decreases
B. increases
C. remains constant
D. increases very fast
The rate of reaction decreases with the passage of time as the concentration of reactants decrease.
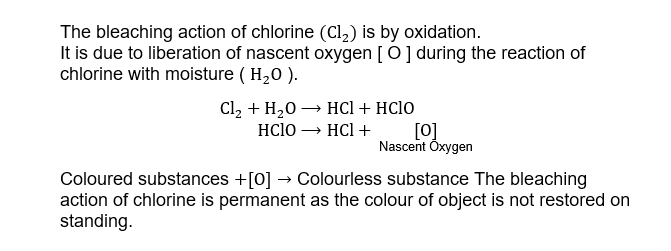
9. Bleaching action of chlorine is by
A. decomposition
B. hydrolysis
C. reduction
D. oxidation
10. Assertion (A) A chemical reaction becomes faster at higher temperatures.
Reason (R) At higher temperatures, molecular motion becomes more rapid.
Codes
A. Both A and R are individually true and R is the correct explanation of A .
B. Both A and R are individually true but R is not a correct explanation of A .
C. A is true but R is false.
D. A is false but R is true.
Both A and R are individually true and R is the correct explanation of A .
A chemical reaction becomes faster at higher temperatures due to increase in molecular motion. When temperature is raised, the kinetic energy of the particles increases. Due to the increase in kinetic energy, the particles start vibrating with greater speed. These particles collide more frequently and hence, rate of chemical reaction increases.
11. Match List I (oxidation number) with List II (the element) and select the correct answer using the codes given below the lists
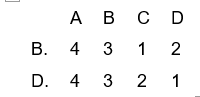
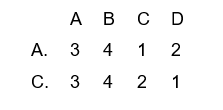
A.
B.
C.
D.

12.Which of the following slows down the reaction rate?
A. Catalytic parameter
B. Homogeneous catalyst
C. Heterogenous catalyst
D. None of the above
All of the given options increase the rate of reaction. Catalysts are substances which reduce the activation energy barrier for any reaction. Thus, catalytic parameter, heterogeneous catalyst and homogeneous catalyst increases the rate of reaction.
13. Which one of the following actions is related with evaporation of sweat?
A. Exothermic action
B. Endothermic action
C. Chemical action
D. Salty action
Endothermic action is related with evaporation of sweat. Sweating allows the body to regulate its temperature. When the external temperature reaches 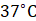 or above, body recognises that the temperature in the surrounding air is higher than the temperature of the body.
or above, body recognises that the temperature in the surrounding air is higher than the temperature of the body.
So, it perspires. The sweat evaporates from the skin using the body heat and making our body a bit cooler.
14. A reaction is spontaneous, when
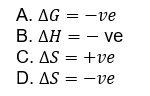
A.
B.
C.
D.
A reaction is spontaneous when 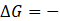 ve that is when Gibbs' free energy change is negative in the reaction, then it can carry out reaction by itself and it is said to be spontaneous.
ve that is when Gibbs' free energy change is negative in the reaction, then it can carry out reaction by itself and it is said to be spontaneous.
15. Example of corrosion is
A. black coating on silver
B. green coating on copper
C. brown coating on iron
D. above three
E. None of these
A.
B.
C.
D.
The gradual destruction of pure metals by the action of air, moisture or a chemical (such as an acid) on their surface is called corrosion. Rusting of iron, black coating on silver, green coating on copper, brown coating on iron are all examples of corrosion.
© 2024 | The Tutors Academy | Website Conceptualised and Developed by Promotion Paradise
