1. Who discovered electrons?
A.Rutherford
B.Niels Bohr
C.J.J. Thomson
D.Newton
J.J. Thomson discovered the electrons while performing cathode ray experiment. During his experiment, he found that these rays consist of negatively charged particles, called electrons.
2. A proton posses what type of charge?
A.Positive
B.Negative
C.No charge
D.Either positive or negative
An atom consist of three main fundamental particles i.e., electrons, protons and neutrons. In which electron is negatively charged, proton is positively charged and neutron has no charge.
3. Constituents of atomic nucleus are
A.electrons and protons
B.electrons and neutrons
C.protons and neutrons
D.protons, neutrons and electrons
Constituents of an atomic nucleus are protons and neutrons and the nucleus of an atom is surrounded by electrons.
4. Consider the following events and arrange them in correct chronological order starting from the earliest to the last activity.
I. Discovery of pi meson
II. Discovery of Neutron
III. Discovery of Electron
IV. Discovery of Proton
Select the correct answer using the codes given below:
A.I, III, IV, II
B.III, IV, II, I
C.III, IV, I, II
D.III, II, I, IV
The correct chronological order is-III, IV, II, I
The pi meson (Pions) was discovered by Japanese physicist Yukawa Hideti in 1935.
5. The atomic nucleus was discovered by
A.Rutherford
B.Dalton
C.Einstein
D.Thomson
The atomic nucleus was discovered by Ernest Rutherford in 1911. Nucleus is a very dense region consisting of protons and neutrons at the centre of an atom.
6. Consider the following statements.
1. An atom is electrically neutral.
2. The negative charge on nucleus is equal to the total negative charge of all the orbiting electrons.
Select the correct answer using the codes given below.
A.Both are incorrect.
B.Both are correct.
C.1 is correct, 2 is incorrect.
D.1 is incorrect, 2 is correct.
Statement (1) is correct, as atom is electrically neutral because the total positive charge (of protons) is cancelled by total negative charge (of electrons). Statement (2) is incorrect because nucleus is positively charged and does not have any negative charge.
7. The positively charged part at the centre of an atom is called as
A.proton
B.neutron
C.electron
D.nucleus
The positively charged part at the centre of an atom is called nucleus. An atomic nuclei consist of positively charged particles (protons) and uncharged particles (neutrons), which makes the nucleus positively charged.
8. Which of the following statement about atomic structure is correct?
A.Neutron and electron are found inside the nucleus and protons revolve around the nucleus.
B.Electron and proton are inside the nucleus and neutron revolve around the nucleus.
C.Proton and neutron are inside the nucleus and electrons revolve around the nucleus.
D.Proton, neutron and electron all are inside the nucleus.
Statement (c) is correct, while statements (a), (b) and (d) are incorrect as only protons and neutrons are inside the nucleus and electrons revolve around the nucleus.
9. Which of the following is not a sub-atomic particle?
A.Neutron
B.Proton
C.Deuteron
D.Electron
Sub-atomic particles aro the particles found inside an atom and are responsible for forming the structure of an atom. Electrons, protons and neutrons are the basic sub-atomic particles while deuteron is not a sub-atomic particle, it is the isotope of hydrogen and it contains one proton and one neutron.
10. Which one of the following pairs constitutes particle-antiparticle pair?
A.Electron-Positron
B.Proton-Neutron
C.Photon-Electron
D.Neutron-Neutrino
The positron (positively charged electron) is the anti-particle of the negatively charged electron. The anti-particle has the same mass and opposite charge that's why only option (a) is a particle-anti particle pair.
11. Which one is a fundamental particle?
A.Neutron
B.Proton
C.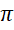 -meson
-meson
D.Quark
Quark is a fundamental particle of matter. They combine to form composite particles called hadrons, the most stable hadrons are protons and neutrons.
12. Higgs boson are also known as
A.electron
B.proton
C.neutrons
D.God particles
The Higgs boson is also known as "God particle" as per the book 'The God Particle' by Nobel Laureate Leon Lederman. It is the fundamental particle associated with the Higgs field, which gives mass to other particles such as electrons and quarks.
13. The nucleus of helium has
A.only one neutron
B.two protons
C.two protons and two neutrons
D.one proton and two neutrons
Atomic number of helium is 2 and atomic mass is 4 . So, there are 2 protons and 2 neutrons in the nucleus of helium.
14. Which of the following elements does not contain neutrons?
A.Oxygen
B.Nitrogen
C.Hydrogen
D.Copper
Hydrogen is the only element that does not contain any neutron in its nucleus. It has only one electron (-ve charged) and one proton (+ve charged). Whereas, oxygen has 8 protons and 8 neutrons, nitrogen has 7 protons and 7 neutrons and copper has 29 protons and 35 neutrons.
15. Which of the following properties of atom could be explained correctly by Thomson model of atom?
A.Overall neutrality of atom
B.Spectra of hydrogen atom
C.Position of electrons, protons and neutrons in atom
D.Stability of atom
Overall neutrality of atom could be explained correctly by Thomson model of atom. As this model states that the positive charge is uniformly distributed in the atom and the electrons are embedded such that atom possess the most stable electrostatic arrangement. Therefore, the model was able to explain the overall neutrality of the atom
© 2024 | The Tutors Academy | Website Conceptualised and Developed by Promotion Paradise
