1. The atoms, in which the number of protons is same but the number of neutrons is different, are known as
A.isobars
B.isomers
C.isotones
D.isotopes
The atoms, in which the number of protons is same but the number of neutrons is different are known as isotopes.
2. Which of the following is not a part of an atom?
A.Electron
B.Proton
C.Neutron
D.Photon
Photon is not a part of an atom. It is a packet of energy, which travels with the velocity of light. All types of electric and magnetic waves occur due to photons, while electrons, protons and neutrons are the main constituents of an atom.
3. In an atom, the order of filling up the orbitals is governed by
A.Aufbau principle
B.Heisenberg's uncertainty principle
C.Hund's rule
D.Pauli's exclusion principle
In an atom, the order of filling up of orbitals is governed by Aufbau principle. It states that, in the ground state of an atom, the electrons first fill the lowest energy orbital before occupying higher energy orbitals.
4. What is the maximum number of electrons in the 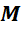 -shell?
-shell?
A.6
B.8
C.18
D.32
The maximum number of electrons in the 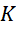 -shell is 18 . According to Bohr, the first orbit is
-shell is 18 . According to Bohr, the first orbit is 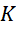 -orbit which occupies a maximum of 2 electrons. The second orbit or the
-orbit which occupies a maximum of 2 electrons. The second orbit or the 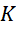 -orbit occupies 8 electrons, the third orbit or the
-orbit occupies 8 electrons, the third orbit or the 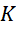 -orbit occupies a maximum of 18 electrons. Likewise the fourth orbit or the
-orbit occupies a maximum of 18 electrons. Likewise the fourth orbit or the 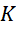 -orbit occupies 32 electrons.
-orbit occupies 32 electrons.
5. The uncertainty principle was proposed by
A.de-Broglie
B.Maxwell
C.Thomson
D.Heisenberg
The uncertainty principle was proposed by Heisenberg. According to this principle, the position and speed of an electron in an orbit cannot be determined simultaneously.
6. Which among the following is an incorrect option?
A.Atom is electrically neutral due to presence of protons and electrons inside the nucleus.
B.Electrons are negatively charged and have negligible mass.
C.Atomic number is the number of protons in an atom of an element.
D.Isotopes are useful as nuclear fuel, in medical field, in carbon dating etc.
Statement (a) is incorrect while statements (b), (c) and (d) are correct.
The corrected form of (a) is as follows;
Atom is electrically neutral as protons and neutrons embedded inside the nucleus and electrons spinning around the nucleus.
7. The number of electrons and neutrons in an element is 18 and 20 , respectively. Its mass number is
A.22
B.2
C.38
D.20

8. The mass number of a nucleus is
A.the sum of the numbers of neutrons and protons
B.the total mass of neutrons and protons
C.always more than the atomic weight
D.always less than its atomic number
The mass number of a nucleus is the sum of the number of neutrons and protons. It is represented by 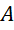 and is different for different elements.
and is different for different elements.

A
B
C
D
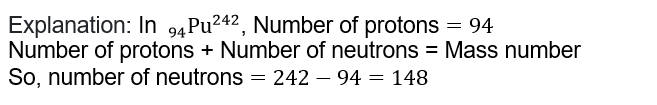
10. Isotopes of an element differ in
A.the number of protons
B.the mass number
C.the number of electrons
D.the atomic number
Isotopes of an element differ in the mass number. They are atoms of the same element that have different numbers of neutrons but the same number of protons and electrons.
11. The maximum kinetic energy of emitted electrons in a photoelectric effect does not depend upon
A.wavelength
B.frequency
C.intensity
D.work function
The maximum kinetic energy of emitted electrons in a photoelectric effect does not depend upon intensity of light which represents the number of photons per unit area. If intensity is increased, more photons strike the surface per unit area, hence more electrons are ejected, which has nothing to do with the kinetic energy of the ejected electrons. However, the kinetic energy of ejected electrons depends upon the wavelength and frequency of light and work function of the surface from which it is emitting.
12. Quantum nature of light is not supported by the phenomenon of
A.compton effect
B.photoelectric effect
C.emission and absorption spectrum
D.interference of light waves
Interference of light waves.
Compton effect, photoelectric effect and emission and absorption spectrum are evidence to show the particle nature of light and hence, the quantum theory of light i.e. energy is absorbed by the electrons of an atom in the form of energy packets called photons.
Whereas, the wave nature of light is exhibited by interference of light waves.
13. What are isotones?
A.Atoms have same number of protons but different number of neutrons.
B.Atoms have same number of neutrons but different number of protons.
C.Atoms have same number of protons and neutrons.
D.Atoms of the same element with different masses.
Atoms that have same number of neutrons but different number of protons are known as isotones.
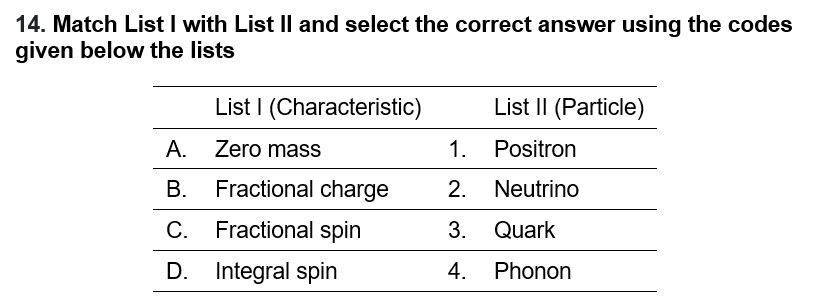
| A2 | B3 | C1 | D4 |
| A3 | B2 | C4 | D1 |
| A2 | B3 | C4 | D1 |
| A3 | B2 | C1 | D4 |

15. The efforts to detect the existence of Higgs-boson particle have become frequent news in the recent past. What is/are the importance of discovering this particle?
3. It will enable us to create better fuel for nuclear fission.
Select the correct answer using the codes given below.
A.Only 1
B.2 and 3
C.1 and 3
D.All of the above
Higgs-boson particle, sometimes also referred as God particle, is a particle associated with the Higgs field, a field which gives mass to other elementary particles. So, it helps us to understand as to why elementary particles have mass.
© 2024 | The Tutors Academy | Website Conceptualised and Developed by Promotion Paradise
