1. The reaction taking place in stars making them shine is
A. nuclear fusion
B. nuclear fission
C. neutron emission
D. gamma decay
Among the given options, the reaction taking place in stars making them shine is nuclear fusion.
2. Which of the following method is used for measuring the age of the earth?
A. Carbon dating process
B. Biotechnology law
C. Biological clock method
D. Uranium process
Uranium process is used for measuring the age of the earth, decaying rocks and bones.
3. Carbon dating is used to determine the age of
A. trees
B. earth
C. fossils
D. rocks
Carbon dating process is used to determine the age of fossils.
4. Which one of the following is incorrect?
A. Theory of evolution was propounded by Charles Darwin.
B. The breaking apart of the nucleus of an atom is called fusion.
C. Dry ice is nothing but solid carbon dioxide.
D. Telephone was invented by Graham Bell.
Among the given statements, statement (b) is incorrect because the breaking apart of the nucleus of an atom is called nuclear fission.
5. Which of the following archaeological samples is adopted to determine the age of firewood bone and shell?
A. Uranium-238
B. Argon-isotopes
C. Carbon-14
D. Strontium-90
Carbon-14 is used to determine the age of firewood bone and shell by the process of radio carbon dating. In the atmosphere carbon-14 is produced in the upper atmosphere by secondary  neutrons, which are the result of reaction of cosmic radiation with atmospheric nitrogen.
neutrons, which are the result of reaction of cosmic radiation with atmospheric nitrogen.
6. 'Radioactive dating' is a technique which can be used to measure?
A. The age of rocks
B. Composition of rocks
C. Colour of rocks
D. Weight of rocks
'Radioactive dating' is a technique used to measure the age of rocks.
7. Harnessing of nuclear energy often causes
A. air pollution
B. water pollution
C. thermal pollution
D. noise pollution
Harnessing of nuclear energy often causes thermal pollution. A common cause of thermal pollution is the use of water as coolant in power plants (thermal as well as nuclear power plants).
8. Natural radioactivity was discovered by
A. Marie Curie
B. Ernest Rutherford
C. Henri Becquerel
D. Enrico Fermi
Natural radioactivity was discovered by French scientist Henri Becquerel in 1896, while working with phosphorescent materials. The SI unit of radioactivity is Becquerel (Bq).
9. Radioactivity is measured by
A. radiometer
B. ammeter
C. magnetometer
D. Geiger-Muller Counter

10. Which among the following is a positively charged particle emitted by a radioactive element?

A.
B.
C.
D.
Alpha rays are positively charged particles consisting of two protons and two neutrons. They are produced during alpha decay when a nucleus has too many protons and too little binding energy to hold them together.
11. When 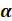 -particle emits from a radioactive element then
-particle emits from a radioactive element then
A. atomic number decreases by 2 and mass number decreases by 4 .
B. atomic number decreases by 1 and mass number decreases by 2 .
C. atomic number increases by 1.
D. atomic number and mass number remains same.
When an -particle is emitted from a radioactive source, its atomic number decreases by 2 and its atomic mass decreases by 4
12. Which of the following metals are used as catalyst in the Catalytic converters to eliminate poisonous gases?
A. Sodium
B. Thorium
C. Rhodium
D. None of these
Rhodium metals are used as catalyst in the Catalytic converters to eliminate poisonous gases. Rhodium is a chemical element, it has symbol Rh and atomic number 45 . It is a very rare, silvery-white, hard, corrosion-resistant transition metal. It is a noble metal and a member of the platinum group. It has only one naturally occurring isotope.
13. Which of the following particles has zero charge?
A. Positron
B. Neutrino
C. Electron
D. 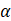 -particle
-particle
Neutrino particle has zero charge. Neutrino is a subatomic particle that is similar to an electron, but it has no electrical charge and has small mass.
14. With reference to the radioactivity, which of the following statements is/are correct?
2. Hydrogen bomb is prepared on the principle of nuclear fission.
Select the correct answer using the codes given below.
A. Only 1
B. Only 2
C. Both 1 and 2
D. Neither 1 nor 2
With reference to the radioactivity, statement 1 is correct. Radioactivity is the process in which the nucleus of an unstable atom loses energy by emitting particles of ionising radiation. Hence, we can say that it is a nuclear property. However, statement 2 is incorrect because hydrogen bomb is based on the principle of uncontrollable nuclear fusion.
15. Which of the following radio isotopes is used in the treatment of blood cancer (leukemia)?
A. Iodine-131
B. Sodium-24
C. Phosphorus-32
D. Cobalt-60
Phosphorus-32 is used to treat blood cancer (leukemia). It is also used to detect tumors.
Iodine-131 is used to treat hyperthyroidism and thyroid cancer.
Sodium-24 is used as a tracer to diagnose various malfunctioning including heart disease.
Cobalt-60 is used to treat malignant tumors which may cause cancer.
© 2024 | The Tutors Academy | Website Conceptualised and Developed by Promotion Paradise
