1.Which is the most common isotope of uranium found in nature?
A. Uranium-235
B. Uranium-238
C. Carbon- 14
D. Rubidium-87
Uranium-238 is the most common isotope of uranium.
Uranium-235 is the second most common isotope and find applications in nuclear reactors and nuclear weapons.
Carbon-14 (isotope of carbon) and rubidium-87 (isotope of rubidium) are not the isotopes of uranium.
2. Radioisotopes are not used in
A. radiocarbon dating
B. making atom bomb
C. cancer therapy
D. purification of drinking water
Radioisotopes are not used in purification of drinking water instead it contaminates water and makes it unsuitable for drinking purpose.
Radiocarbon dating involves C-14 isotope to determine the age of an object containing organic material.
Atom bomb mostly consists of uranium-235 and plutonium-239 isotopes.
Cancer therapy involves cobalt-60, phosphorus-32, etc for treatment of different types of cancer.
3. The half-life of a radioactive substance is 4 months. How much time it will take to decay 3/4th of the substance?
A. 12 months
B. 8 months
C. 4 months
D. 3 months
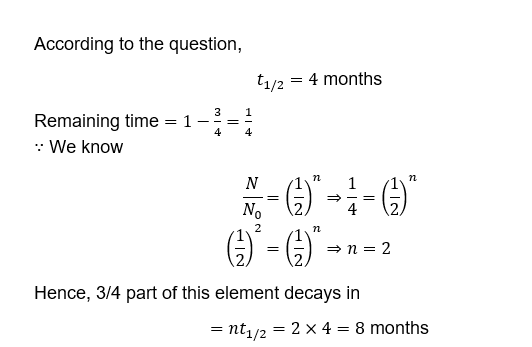
4. A team of scientists at Brookhaven National Laboratory including those from India created the heaviest anti-matter (anti-helium nucleus). What is/are the implication/implications of the creation of anti-matter?
A. Only 1
B. 2 and 3
C. Only 3
D. 1,2 and 3
Statements (2) and (3) are correct.
Anti-matter particles share the same mass as their matter counter parts, but properties such as electric charge are opposite. Anti-helium-4 is a conglomeration of two antiprotons and two antineutrons. This antihelium is the antimatter to alpha-particle. Its discovery will help us to understand the evolution of the universe. It will also help to probe the possibility of the existence of stars and galaxies made of anti-matter.
5. With reference to the isotopes of elements, consider the following statements
A. The isotopes of cobalt are used in the treatment of cancer.
B. The isotopes of iron are used in the diagnosis of anemia.
C. The isotopes of uranium are used as fuel in nuclear reactors.
D. The isotopes of iodine are used in the treatment of Hepatitis A.
Select the correct answer using the codes given below.
A. A, B and C
B. A and B
C. A, B, C and D
D. B and C
Statements 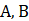 and C are correct. However Statement D is incorrect, as the isotopes of iodine are used in the treatment of goitre and thyroid.
and C are correct. However Statement D is incorrect, as the isotopes of iodine are used in the treatment of goitre and thyroid.
6. Consider the following statements.
A. Nuclear fuel is material used in nuclear power stations to produce heat to run turbines.
B. Thorium is used as a nuclear fuel in India.
Which of the above statements is/are correct?
A. Only A
B. Only B
C. Both A and B
D. Neither A nor B
The nuclear fuel is used in nuclear power stations to produce heat to power turbines. Heat is produced when nuclear fuel undergoes nuclear fission.
Thorium fuel is used as a nuclear fuel in India.
7. The isotope of carbon used in radiocarbon dating is
A. C - 12
B. C -13
C. C - 14
D. C - 15
Among the given options, the isotope of carbon used in radiocarbon dating is C-14.
8. Change of water into the vapour is called
A. natural
B. physical
C. chemical
D. biological
A change in which a substance undergoes change in its physical properties such as shape, size, etc. is called a physical change. Change of water into the vapour form is a physical change.
9. An example of physical change
A. blackening of silverware
B. burning of candle
C. making of curd from milk
D. dissolving of sugar in water
Dissolving of sugar in water is an example of physical change. A physical change is generally reversible. However, other options like blackening of silverware, burning of candle and making of curd from milk are examples of chemical changes.
10. If an unsaturated sugar solution is prepared by mixing 100 gm of sugar with half-litre of water. Which of the following physical quantities will not change?
A. Mass
B. Density
C. Concentration
D. Volume
Volume of the solution will not change when unsaturated sugar solution is prepared by mixing 100 gm of sugar with half-litre of water. It is because, empty spaces are present between the molecules of liquid in unsaturated solution. So, the sugar molecules do not occupy new spaces instead they get dissolved and occupy those empty spaces.
11. Which of the following is an example of chemical change?
A. Dispersion of white light into the light of seven colours when passed through a prism.
B. Softening of vegetables when cooked.
C. Wet soil lamp when dried gets fragile.
D. Dissolving of salt in water.
Softening of vegetables when cooked is an example of chemical change. A chemical process is a process in which a substance that takes part in it cannot return to its original state. It is permanent and irreversible.
12. Which of the following is/are the example/ examples of chemical change?
Select the correct answer using the codes given below.
A. 1 and 2
B. Only 3
C. 1,2 and 3
D. None of these
Only souring of milk is an example of a chemical change. This change is permanent and irreversible.
However, crystallisation of sodium chloride and melting of ice are the examples of a physical change.
13. The temperature point at which solid, liquid and gaseous states may stay together is known as
A. boiling point
B. melting point
C. freezing point
D. triple point
E. None of the above/More than one of the above
A.
B.
C.
D.
The temperature point at which solid, liquid and gaseous states may stay together is known as triple point.
14. Detect the odd one.
A. Setting of cement
B. Burning of coal
C. Ripening of fruit
D. More than one of these
E. None of these
A.
B.
C.
D.
All the given processes are examples of a chemical change. Thus, none of the given option is odd one out.
15. A substance consisting of two or more ingredients which are not in chemical combination is called
A. an ion
B. a mixture
C. a molecule
D. an oxide
Substance that consists of two or more ingredients which are not in chemical combination is called as a mixture.
In mixtures, the ingredients are not chemically combined and can be separated by simple physical processes like filtration, boiling etc.
© 2024 | The Tutors Academy | Website Conceptualised and Developed by Promotion Paradise
