1.Given below are two statements, one is labelled as Assertion (A) and the other as Reason (R).
Assertion (A) A solution of table salt in a glass of water is homogeneous.
Reason (R) A solution having different compositions throughout is homogeneous.
Select the correct options given below.
A. Both A and R are true and R is the correct explanation of A .
B. Both A and R are true, but R is not the correct explanation of A .
C. A is true, but R is false.
D. A is false, but R is true.
Assertion 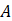 is true, but reason
is true, but reason 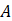 is false because salt water is a homogeneous mixture. It dissolves completely in water and dissociates into sodium and chloride ions.
is false because salt water is a homogeneous mixture. It dissolves completely in water and dissociates into sodium and chloride ions.
Homogeneous solutions are those solutions which have same composition and properties throughout the mixture.
e.g. Solution of salt or sugar in water.
2. Given below are two statements.
Statement I Rusting of Iron is a chemical change. Statement II The process of depositing a layer of zinc on iron is called galvanisation.
In the light of the above statements, choose the most appropriate answer from the options given below:
A. Both Statement I and Statement II are correct
B. Both Statement I and Statement II are incorrect
C. Statement I is correct and Statement II is incorrect
D. Statement I is incorrect and Statement II is correct
Both statement 1 and statement II are correct.
Rusting of iron involves the formation of iron oxide through the reaction of iron with oxygen and water, which is a chemical change. Galvanisation is the process of applying a protective zinc coating to iron or steel to prevent rusting.
3.Milk is an example of
A. a gel
B. a foam
C. an emulsion
D. a suspension
Milk is an emulsion of fat and water, along with other components. In an emulsion, one liquid (the dispersed phase) is dispersed in the other (the continous phase).
4. Which of these is not a colloid?
A. Milk
B. Blood
C. Ice-cream
D. Honey
Honey is not a colloid, because in colloidal solution the solute particles are larger in size and are able to show the "Tyndall effect". Milk, blood and ice-cream have solute particles which are larger in size and they show "Tyndall effect". Therefore milk, blood and ice-cream are colloids.
5. Indicate which of the following is a pure substance?
Codes
A. 2, 3 and 4
B. Only 1
C. Only 2
D. Only 3
Among the given options, only iron is a pure substance.
Pure substances are the one which are made up of only one type of particles, e.g. all the elements (iron, sulphur, sodium, carbon etc.) and all the compounds (distilled water, ethanol, sulphuric acid etc.)
Rest of the substances are formed as a mixture of two or more pure substances.
6. A mixture of sand and naphthalene can be separated by
A. sublimation
B. distillation
C. chromatography
D. fractional distillation
E. None of these / More than one of these
A.
B.
C.
D.
Mixture of sand and naphthalene can be separated by sublimation. It is because naphthalene displays the phenomenon of sublimation.
7. The conversion of a solid directly into gas is called as
A. sublimation
B. condensation
C. evaporation
D. boiling
E. None of these/More than one of these
A.
B.
C.
D.
Conversion of a solid state directly into its gaseous state is called as sublimation.
Condensation is a process by which a substance changes from a gaseous state to a liquid state.
Evaporation is a process by which liquid changes into gas. It is opposite phenomenon of condensation.
Boiling is the rapid vaporisation of a liquid, which occurs when a liquid is heated to its boiling point (i.e. the temperature at which the vapour pressure of the liquid is equal to the pressure exerted on the liquid by surrounding atmosphere).
8. Process of solid camphor into camphor vapour is called
A. vaporisation
B. freezing
C. melting
D. sublimation
Process of converting solid camphor into camphor vapour is called sublimation. It is the process in which a solid changes directly into vapour.
9. A sample of any of the following substances disappears after sometime when exposed to air like camphor, naphthalene or dry ice. This phenomenon is called
A. sublimation
B. evaporation
C. diffusion
D. radiation
Sublimation process is used to separate two solids in which one of the solids sublimates easily such as naphthalene, camphor or dry ice.
10. Consider the following statements.
In the reverse osmosis, external pressure is applied to the dilute solution.
Which of the statements given above is/are correct?
A. Only 1
B. Only 2
C. Both 1 and 2
D. Neither 1 nor 2
Both the statements (1) and (2) are incorrect because osmosis is the process in which the solvent particles move across a semi-permeable membrane from a dilute solution to a concentrated solution. while, in reverse osmosis, the external pressure is applied to the concentrated solution.
11. After cutting cucumber if salt is added, then water comes out due to
A. active transport
B. passive transport
C. osmosis
D. diffusion
After cutting cucumber if salt is added, then water comes out due to osmosis.
It is a process by which the molecules of a solvent travels from a solution of low concentration to a solution of high concentration through a semi-permeable membrane.
12. Water passes from the soil into the roots by a physical process called
A. diffusion
B. transpiration
C. absorption
D. osmosis
E. None of these/More than one of these
A.
B.
C.
D.
The root hairs take water from the soil by a process called osmosis.
In osmosis, water molecules move from a region of lower concentration of solute to a region of higher concentration of solute through a semi-permeable membrane.
13. Which one of the following process is not useful for purification of solid?
A. Distillation
B. Sublimation
C. Crystallisation
D. All of the above
Distillation is not useful for purification of solid impurities. It is generally used for purification of liquid impurities. While sublimation and crystallisation are used for purification of solid impurities.
14. Consider the following pairs.
I. Ginning: Separation of cotton fibres from seeds
II. Weaving : Making yarn from fibres
III. Polyester : Melts on catching fire and sticks to the body
How many of the above pairs are correctly matched?
A. Only one pair
B. Only two pairs
C. All three pairs
D. None of the pairs
E. Question not attempted
A.
B.
C.
D.
Only two pairs are correctly matched i.e., pair (I) and (III).
Ginning is the process by which the fibers are separated from the seeds.
Polyester, a type of synthetic fabric, does melt when exposed to fire and can stick to the body. The process of making yarn from fibres is called spinning.
Pair (II) is incorrect matched.
Weaving is the textile art in which two distinct sets of yarns or threads called the warp and weft are interlaced with each other at right angles to form a fabric or cloth. The warp threads run length ways on the piece of cloth, while the weft runs horizontally.
15. Pasteurisation is a process by which
A. milk is preserved at a very low temperature for 24 hours.
B. milk is heated for 8 hours.
C. first milk is heated up to a longer time and then cooled suddenly within specified time.
D. None of the above
Pasteurisation is a process by which, first milk is heated up to a longer time and then cooled suddenly within the specified time. It is also known as sterilisation of milk.
© 2024 | The Tutors Academy | Website Conceptualised and Developed by Promotion Paradise
